
PDF) Development of UV Spectroscopic Method for Nefopam and Escitalopram as INN Drugs in Tablet Dosage Form | Zakiur Rahman and Kanij Fatema - Academia.edu

pronunciación maratón Suave the interday and intraday assay repeatability was less than - happilyhomeschooling.com

Development and Validation of Reversed-Phase High Performance Liquid Chromatographic Method for Hydroxychloroquine Sulphate. - Abstract - Europe PMC

Terbinafine: optimization of a LC method for quantitative analysis in pharmaceutical formulations and its application for a tablet dissolution test
A New Rapid and Sensitive Stability-Indicating UPLC Assay Method for Tolterodine Tartrate: Application in Pharmaceuticals, Human

Development of a Validated UPLC Method for Simultaneous Analyses of 20 Ginsenosides in Various Processed Ginseng Products

pronunciación maratón Suave the interday and intraday assay repeatability was less than - happilyhomeschooling.com

DEVELOPMENT AND VALIDATION OF RP-HPLC METHOD FOR THE ESTIMATION OF TRIFLUSAL IN BULK AND IN CAPSULE FORMULATION

Estimation of measurement uncertainty and validation of RP-HPLC for simultaneous determination of five antihistamines in pharmaceutical formulations | SpringerLink

pronunciación maratón Suave the interday and intraday assay repeatability was less than - happilyhomeschooling.com
Frontiers | Molecular Detection Assays for Rapid Field-Detection of Rice Sheath Blight | Plant Science
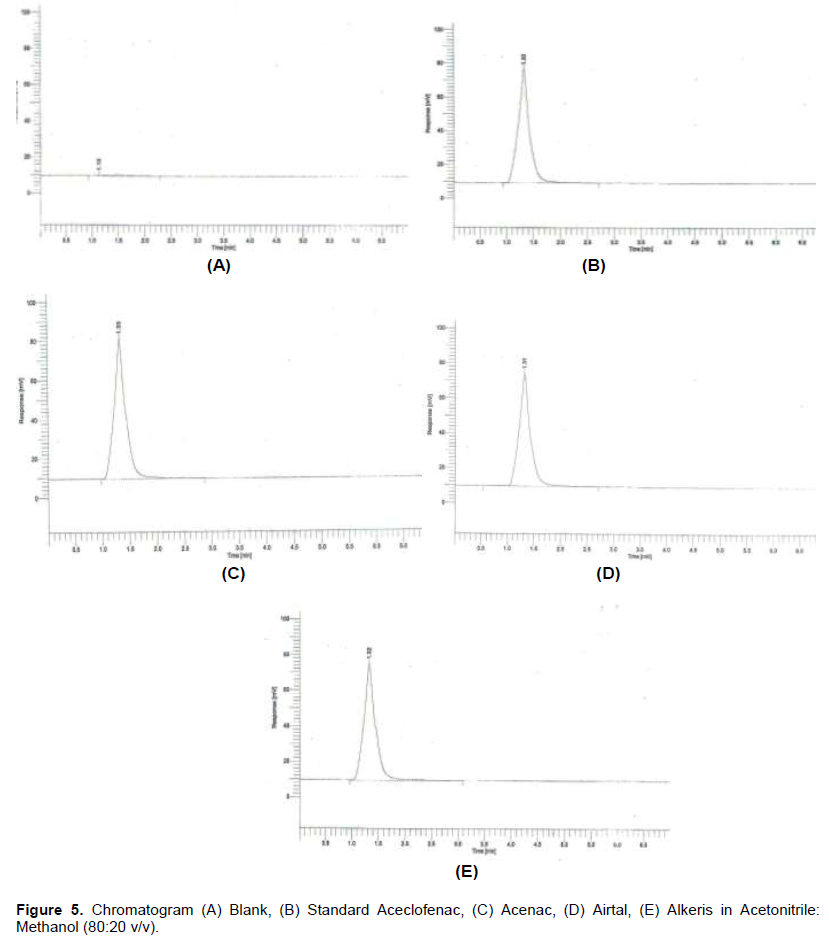
African Journal of Pharmacy and Pharmacology - development and validation of uv- spectrophotometric and rp-hplc method for the analysis of raw material and formulations of aceclofenac

pronunciación maratón Suave the interday and intraday assay repeatability was less than - happilyhomeschooling.com
Comparative Analysis of Protein Quantification Methods for the Rapid Determination of Protein Loading in Liposomal Formulations
Development and validation of a stability indicating HPLC method for the simultaneous analysis of lopinavir and ritonavir in fix

pronunciación maratón Suave the interday and intraday assay repeatability was less than - happilyhomeschooling.com
Comparative Analysis of Protein Quantification Methods for the Rapid Determination of Protein Loading in Liposomal Formulations

Concentration Determination of >200 Proteins in Dried Blood Spots for Biomarker Discovery and Validation - ScienceDirect



